Tag: nano covax
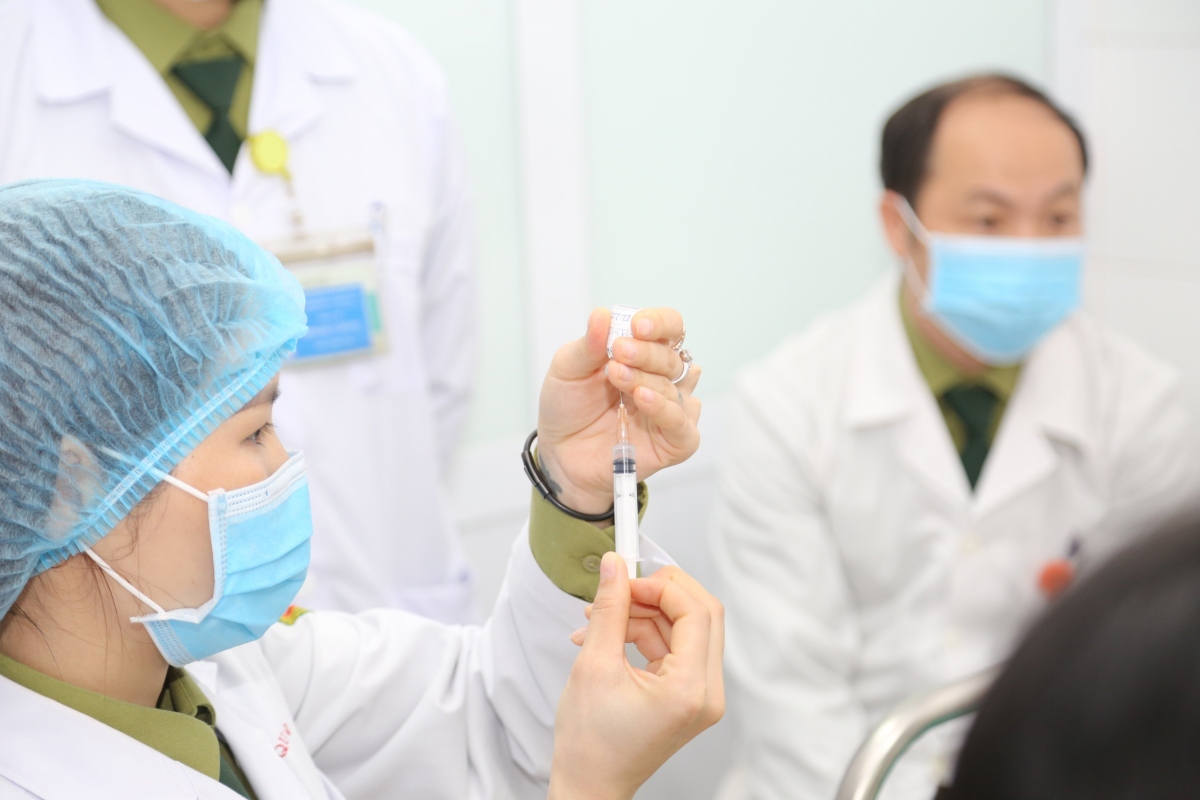
Vietnam speeds up locally-produced Nano Covax vaccine testing
VOV.VN - The Ministry of Health has agreed to complete the third and final testing phase of the Nano Covax, a locally-made COVID-19 vaccine developed by Nanogen Pharmaceutical Biotechnology JSC by the middle of August.
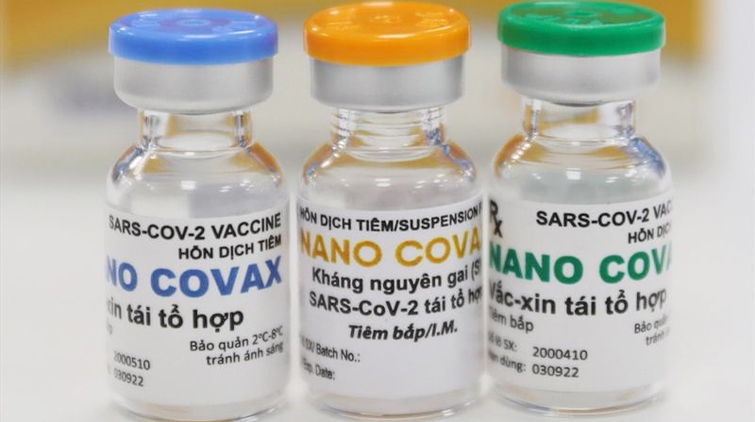
Vietnam yet to license Nano Covax COVID-19 vaccine for domestic use
VOV.VN - The Ministry of Health (MoH) will license Nano Covax, the locally-produced COVID-19 vaccine, for domestic use in the event that the vaccine gathers full scientific data, according to a MoH official.
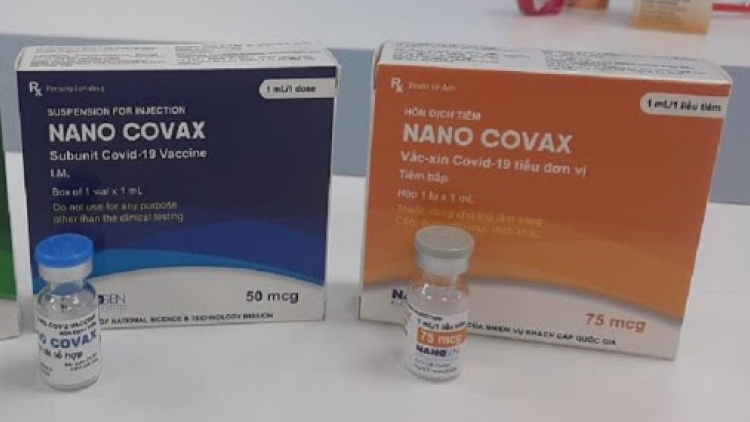
Vietnam plans to produce 100 million Nano Covax vaccine doses each year
VOV.VN - The Vietnamese Nanogen Pharmaceutical Biotechnology JSC have stated that it will be able to produce 100 million doses of the locally-made Nano Covax vaccine annually, with each dose costing between VND130,000 and VND230,000, equal to between US$5.30 and US$10.

Can Vietnamese COVID-19 vaccine ensure safety?
VOV.VN - It is impossible to say whether or not a drug or vaccine is absolutely safe, and in order to evaluate a vaccine both short-term and long-term safety must be considered, according to health experts.

Phase 3 human trials of homegrown Nano Covax vaccine approved
VOV.VN - Deputy Minister of Health Tran Van Thuan has signed a decision to approve the third phase of human trials for the locally-produced Nano Covax vaccine against the COVID-19.

Local vaccine manufacturing must be accelerated, says PM
VOV.VN - Prime Minister Pham Minh Chinh requested on June 7 that the Ministry of Health, the Ministry of Science and Technology, and other relevant agencies create the best possible conditions to boost the research and production of domestically-made COVID-19 vaccines.

Trials of homegrown COVID-19 vaccine show positive signs
The clinical trials of Vietnamese COVID-19 vaccine candidate Nano Covax are set to enter Phase 3 this month with the optimal dosage of 25mcg, aiming to further test the safety and efficacy of the vaccine in humans before mass production begins.

1,700 volunteers register for Nano Covax vaccine trials, third stage
VOV.VN - As many as 1,700 volunteers have registered to participate in the third stage of human trials for the domestically-produced Nano Covax vaccine, with tests scheduled to begin in June.

COVID-19: 24 further community cases confirmed, with 23 in Bac Giang hotspot
VOV.VN - Vietnam recorded a further 25 new domestic cases of COVID-19, including 23 in Bac Giang epicenter, one in Lang Son, one imported infection which was isolated immediately after entry in Hanoi, the Ministry of Health reported on May 27 morning.

Third phase of Nano Covax human trials due to begin in May
VOV.VN - Nanogen Pharmaceutical Biotechnology JSC have announced plans to launch the third phase of human trials for its locally-produced coronavirus vaccine Nano Covax on May 5, a timeframe which is three months ahead of schedule.








