Vietnam trials locally made coronavirus vaccine on people today
VOV.VN - First human trials of Made-in-Vietnam for COVID-19 are set to begin at the Military Medical Academy in Hanoi on December 17.
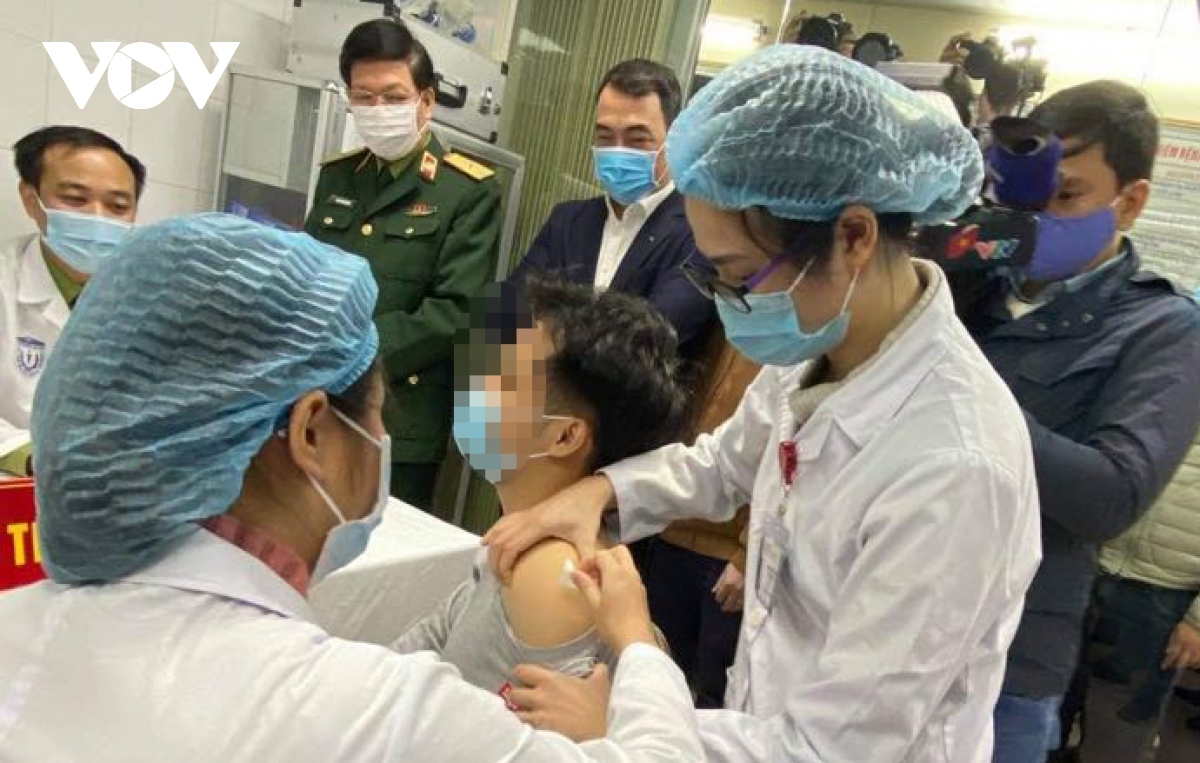
The vaccine, Nano Covax, produced by Nanogen Pharmaceutical Biotechnology JSC, has been transferred to Hanoi for the clinical testing campaign.
More than 200 people have signed up for the testing and Nanogen has also purchased insurance for the volunteers.
In the first phase, 60 selected people aged 18-50 will get Nano Covax shots to test the vaccine’s safety. They will be divided into three groups of 20 people to be injected twice for three corresponding doses of 25, 50 and 75mcg.
Three people of the 25mcg group will be vaccinated first on Dec. 17.
The volunteers will get the second shot after four weeks.
Following the injections, they will be monitored at the Military Medical Academy over 72 hours to see if there are any reactions. They will also be monitored in several months later to evaluate the safety of the vaccine.
Nanogen researchers say the Nano Covax vaccine has undergone rigorous pre-clinical safety tests. It has been shown to be safe and produced encouraging signs of an effective immune response in animal studies.
Nanogen is one of the four COVID-19 vaccine producers in Vietnam, alongside Vabiotech, Ivac and Polyvac. Each dose of Nano Covax costs approximately VND120,000 and each person needs two shots of the vaccine to create immunity against COVID-19.




