HFMD cases surge in Vietnam, Health Ministry calls for prompt action
VOV.VN - The Ministry of Health (MoH) has issued an urgent directive to strengthen patient triage, treatment capacity and infection control across the healthcare system, as cases of hand, foot and mouth disease (HFMD) are rising rapidly, particularly in the southern region.
In an official dispatch released on March 31, the MoH called on all medical facilities to enhance preparedness in order to minimise severe cases and fatalities.
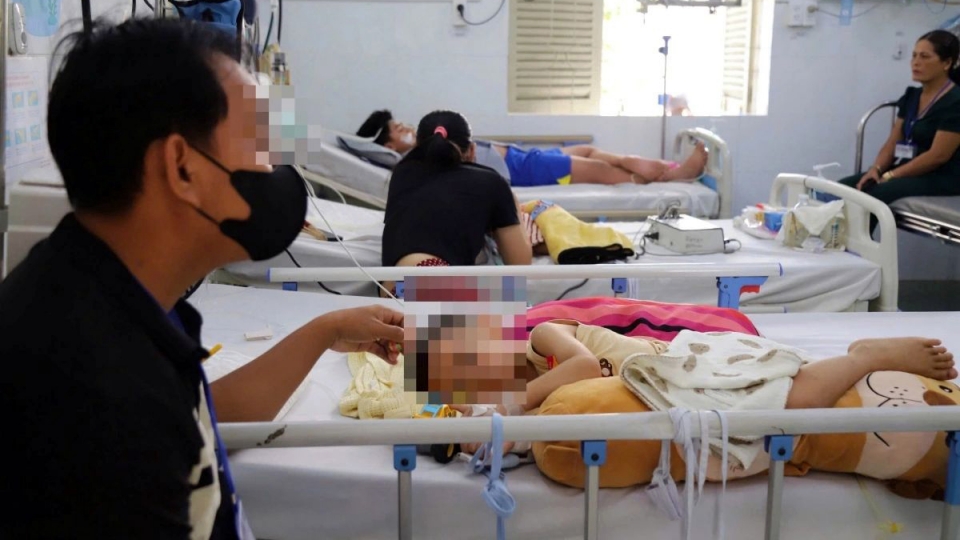
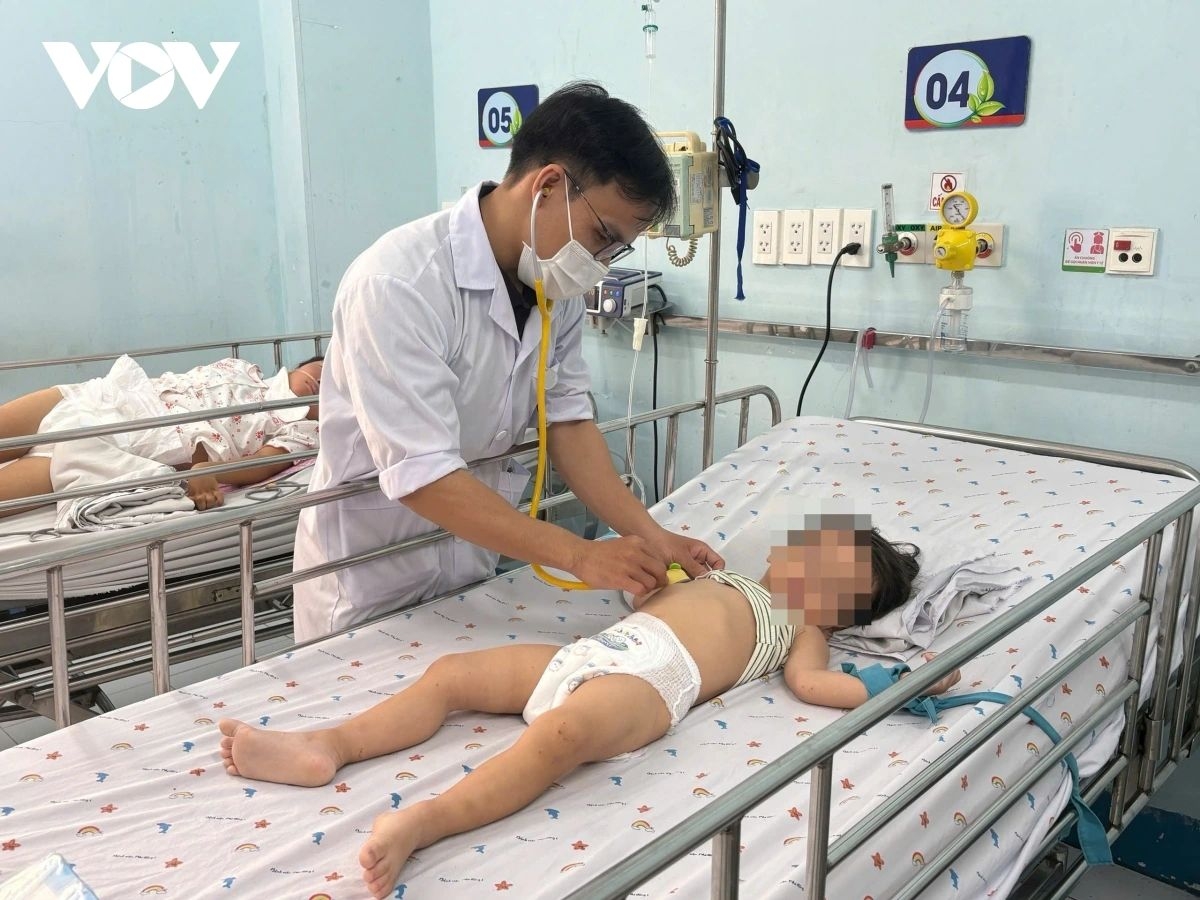
According to the ministry, more than 25,000 HFMD cases have been recorded nationwide in the first three months of 2026, including deaths. Notably, the southern region accounts for nearly 72% of total infections, with over 18,000 cases reported. The disease primarily affects children aged 1–5.
Hospitals, especially major pediatric facilities in southern Vietnam, have reported a sharp increase in outpatient visits and hospital admissions, including a number of severe cases. Surveillance data indicates the circulation of Enterovirus 71 (EV71), a highly virulent strain that can cause serious neurological complications and increase the risk of death.
In response, the MoH has instructed healthcare units to urgently review and update their disease prevention and control plans, while preparing for potential surges in cases. Facilities are required to ensure readiness in patient reception, emergency care, isolation and treatment.
Public communication has been identified as a key priority, aiming to raise awareness among parents and caregivers about transmission routes, early symptoms and preventive measures.
Medical facilities are also required to ensure sufficient staffing, medicines and equipment in line with current treatment guidelines, while strengthening training to improve early detection of severe symptoms, particularly at the grassroots healthcare level. Hospitals are urged to reinforce intensive care capacity and ensure timely referrals for severe cases.
The ministry also stressed the need for close coordination with the preventive health system in epidemiological surveillance, along with stricter infection control measures within hospitals to prevent cross-infection and the emergence of outbreaks in medical facilities.
Infectious disease reporting must be conducted fully and promptly in accordance with regulations.
Regarding vaccination, the Drug Administration of Vietnam on March 17 approved 13 vaccines and biological products for new registration or renewal, including the Envacgen vaccine for HFMD, which contains an inactivated Enterovirus A71 (EV71) component.
Although registered by a Taiwan-based company, the vaccine underwent Phase 3 clinical trials in Vietnam in collaboration with the Pasteur Institute in Ho Chi Minh City. Results show a high protective efficacy against the EV71 strain circulating in Vietnam and Taiwan (China), reducing the risk of infection at all severity levels by up to 96.8%.
Notably, no HFMD cases were recorded among vaccinated children during a two-year follow-up period. In terms of safety, adverse reaction rates were similar between the vaccine and placebo groups, with most reactions being mild and self-resolving, and no serious vaccine-related events reported.
Despite receiving regulatory approval, the HFMD vaccine has not yet been deployed. Authorities expect it to be imported into Vietnam between September and October 2026.